Trouble finding a product?
Contact us.
Desiccators
Desiccators use desiccant to protect container and enclosure contents from moisture damage.
- Container Access
- Corrosion Prevention
- Humidity Monitoring
- Moisture Control
- Moisture Purging
- Pressure Control
- Shelving & Straps
- Shock Monitoring
Find What You're Looking For
It’s a lot, we know. If you’re looking for something specific or aren’t sure what you need, try searching through all products or give us a shout and we’ll help!
What are desiccators?
Desiccators (also known as dehydrators or dehumidifiers) are cannisters or tubes that hold desiccant and function as self-contained moisture control systems. Air is allowed to flow through the desiccator (and thus must flow through the desiccant), before entering an enclosure to protect the contents inside from moisture damage and are used in a wide variety of aerospace and electronic applications.
Types of dessicators
There are two basic types of desiccators: dynamic and static.
Dynamic Desiccators allow a moist gas stream to flow through the desiccant bed to dry the gas. More complicated types of dynamic desiccators are self-regenerating, usually large and expensive, and require an external power source.
Static Desiccators, on the other hand, can vary in size from tiny packets or capsules to 55-gallon drum-sized canisters and larger, filled with desiccant. They are generally much less expensive, but require periodic re-generation or replacement.
Over the last 40+ years, we’ve designed and produced hundreds of different static desiccators. The ones shown on this website demonstrate the wide variety of shapes and sizes and manufacturing technologies that are used to fit the required application. They are available in both standard and custom configurations, made from various materials and can be supplied with any of the three principal commonly commercially available desiccants:
- Silica gel
- Molecular sieve
- Bagged clay (used with TA476 and TA486 desiccant holders and baskets).
Optional features, such as breather valves, humidity indicators, non-dusting filters, and RFI/EMI shielding can also be incorporated.
How do desiccators work?
The desiccant inside of a desiccator adsorbs moisture from the air inside of the container in which the desiccator is installed. The desiccator may optionally use a humidity indicator or breather breather valve at its head to allow external monitoring of the internal humidity level or pressure relief of the container.
Sizing & Specifications
We’ve made desiccators in literally hundreds of sizes ranging from the size of about a thumbtack and up to eight-foot-long tubes.
Our desiccators can be made from:
- Aluminum
- Stainless steel
- Bronze
- Plastics (including polystyrene)
- ABS
- Polycarbonate
- Acrylic
- Polyester
- Polyproplene
- A combination of plastics and metals
Manufacturing processes for desiccator components can utilize one or more of the following: milling, turning, stamping, vacuum forming, injection molding and casting.
Contact us for more information about custom desiccators.
How To Select the Right Desiccator
For a quick recommendation for a standard desiccator, existing custom desiccator, or to design a new desiccator, we’ll need the following information:
- The volume of interior space to be protected.
- The amount and shape of space available for the desiccator.
- The level of moisture protection required.
- The ambient moisture and temperature levels.
- The length of time desired before recharging.
- The anticipated leakage rate of the enclosure.
- Whether a Breather Valve and/or Humidity Indicator are required.
- Dusting protection required (if any).
- RFI/EMI protection required (if any).
- Any unusual environmental conditions.
Since some of the criteria above are performance requirements and others are time and space limitations, it may be necessary to make compromises that meet realistic objectives. The earlier our Engineers can be involved in the design process, the better we can meet the requirements — contact us to get started today!
Quality Control Considerations
Because both silica gel and molecular sieve react rapidly to moisture, they must be handled under controlled minimum humidity conditions to retain maximum adsorption capacity. All of our desiccators are filled in a dry box and packaged in materials with extremely low water vapor transmission rates.
We caution you to leave the desiccators in their original “as received” packaging (with overpack included) until ready for use to assure maximum adsorbent capacity. In addition, you should establish procedures that will minimize the time between the removal of the product from its package and its installation in the sealed enclosure. It is also important that the work be done under optimal ambient conditions. Both silica gel and molecular sieve can lose up to 5% of their adsorbent capacity in one hour when exposed to 60% RH at an ambient temperature of 86°F (30°C). Reducing ambient conditions to 10% RH at 77°F (25°C) will increase this time period for molecular sieve to 12 hours and for silica gel to 35 hours.
Industry Applications
Desiccators are used in a wide variety of applications including:
- Aerospace
- Shipping & Logistics
- Defense
Contact us to find out if our desiccators are the right solution for your application.
Custom Engineering Service
No two jobs are the same so get the solution that’s right for you. Work with AGM Engineering and get a solution specific to your challenge today.
Where Does Moisture Come From?
(A) TRAPPED MOISTURE is the amount of moisture trapped in an enclosure when it is sealed. There are two sources: water vapor in the air inside the enclosure when it is sealed and water on and inside the solid material contents of the enclosure commonly referred to as hygroscopic moisture. Electronic components are well known for the amount of moisture they will hold in solid solution (hygroscopically). To calculate the amount of moisture in the air, multiply the volume of the enclosure in cubic ft. by the amount of moisture that a cubic ft. of trapped air will hold at a given temperature and relative humidity.
Chart 1 displays the amount of moisture that a cubic ft. of air will hold at sea level at various temperatures and relative humidities. Note that the absolute amount of moisture in saturated air increases exponentially with increasing temperature.
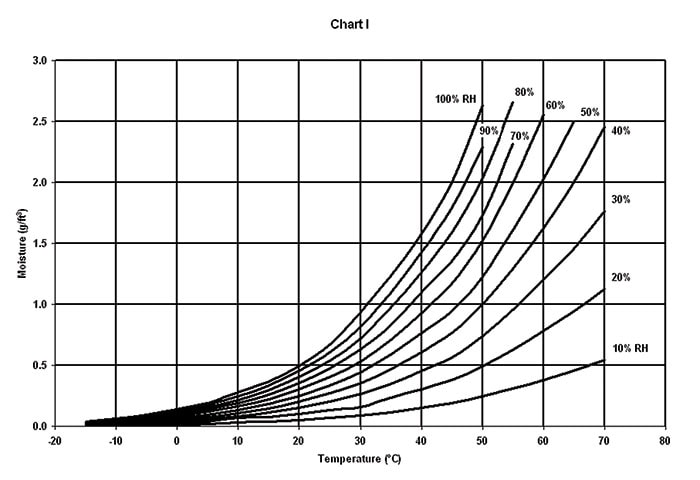
(B) MOISTURE LEAKAGE is the amount of moisture that may enter non-sealed, leaking or porous enclosures as a result of temperature or altitude changes and is directly proportional to the enclosure volume and the differences in temperature or altitude extremes (Chart 2 and Chart 3).
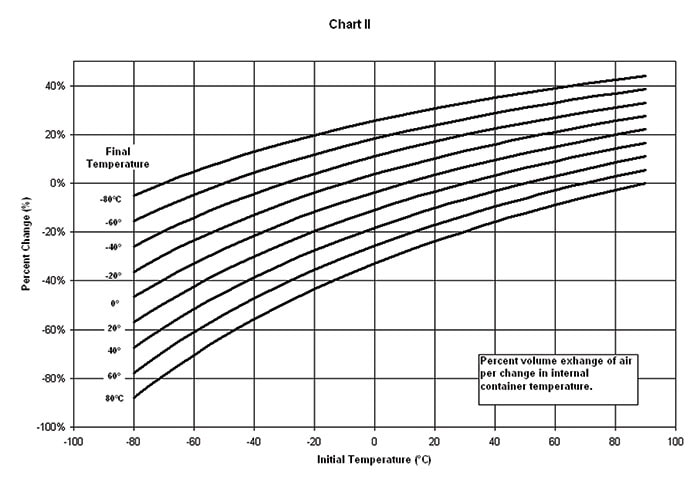
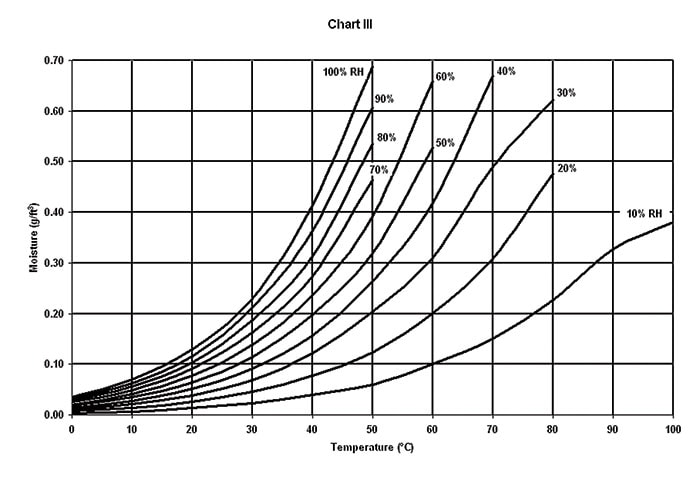
(C) VALVE BREATHING a breather valve’s primary function is to protect an enclosure from excessive pressure or vacuum differentials. Each time the valve opens inward (or “gulps”) it can take in as much as 0.015 g of water per cubic ft. under worldwide storage conditions. During airlift, it can take in as much as 0.09 g of water per cubic ft. during descent. The higher the valve pressure and vacuum reseal settings, the less often the valve will breathe. For this to be an advantage, however, the enclosure must be structurally strong enough to withstand the pressure and vacuum differentials imposed by the valve.
(D) PERMEATION is the movement of individual molecules through a seemingly solid material. Common permeable materials are most seals and seal materials, plastics and other intentionally porous materials. Permeation constitutes a small amount of moisture but over time it can, and will, build up to levels high enough to cause a problem on the inside of the enclosure.
Glossary Terms
The ratio of the mass of water vapor in a sample of air to the mass of air of the sample.
The pressure above the absolute zero value of pressure that is theoretically obtained in empty space. (Example: The absolute pressure at sea level is 14.7 psia.)
The penetration of a substance into the inner structure of a solid or liquid where it is held in place by surface tension effects and is readily displaced.
The surface retention of solid, liquid or gas molecules, atoms or ions by a solid or liquid. The adsorbed material is held in place by Van der Waals and other weak inter-molecular forces and is not readily displaced.
Height, measured as distance along the extended earth’s radius above a given datum, such as average sea level.
A unit of length, 10-8 cm (10-10 meter). Used to define pore size in molecular sieves.
Motion of an aircraft in which the path is inclined upward with respect to the horizontal.
A unit of pressure equal to the pressure exerted by a column of air one inch square and extending from sea level to infinity. 1 atmosphere = 14.7 psi.
Movement of gas, vapors or air in and out of an enclosure as a result of expansions and contractions induced by temperature or pressure change (such as altitude change).
The process by which vapor becomes a liquid and energy is released.
Motion of an aircraft in which the path is inclined downward with respect to the horizontal.
The temperature at which air becomes saturated when cooled without addition of moisture or change of pressure; any further cooling causes condensation.
Pertaining to meteorological actions which are completed within 24 hours and which recur every 24 hours.
An ordinary thermometer, especially one with an un-moistened bulb; not dependent upon atmospheric humidity.
Cushioning material in a container to protect shipped goods from damage.
Condition in which no change occurs in the state of a system as long as its surroundings are unaltered.
A phase of matter in which the substance expands readily to fill any containing vessel; characterized by relatively low density.
A unit of mass equal to 1/7,000 of a pound, or to 6.47989 x 10-5 kilogram (kg).
The unit of mass in the centimeter-gram-second system of units, equal to 0.001 kg.
Water, in any phase, solid (ice), liquid (water), or gas (water vapor).
Having an affinity for, attracting, adsorbing or absorbing water.
Lacking an affinity for, repelling, or failing to adsorb or absorb water.
Atmospheric water vapor content.
Readily absorbs or adsorbs moisture and retains it.
Undesired and gradual escape or entry of a quantity, such as air or water vapor from an enclosure.
A unit of measure of length or distance equal to 1 x 10-6 meters, or 0.00003937 inches.
Water that is dispersed through a gas or liquid in the form of water vapor or small droplets, dispersed through a solid or condensed on the surface of a solid.
A type of stress which is exerted uniformly in all directions; its measure is the force exerted per unit area.
A graph, each point of which represents a specific condition of a gas-vapor system (such as air and water vapor) with regard to temperature (horizontal scale) and absolute humidity (vertical scale); other characteristics of the system, such as relative humidity, wet-bulb or dew point temperature, specific volume and enthalpy, are indicated by lines on the chart.
The act or process of cleaning and purifying by replacing the current contents with a cleaner or purer medium.
The restoring (through the application of heat or other processes) of the adsorption capacity of a desiccant.
The ratio of the actual water vapor pressure of the air-water vapor mixture to the saturation water vapor pressure, stated as a percent (dimensionless) and varies based on temperature.
The point where no more of a substance can be absorbed, adsorbed, dissolved or retained. When referred to desiccant, saturation occurs when the desiccant material will no longer take on moisture for the condition it is exposed to. For water vapor in air, when saturation occurs, liquid water precipitates (or rains) out of the air-water vapor mixture.
A measure of the internal energy of a substance, usually referred to as the degree of hotness or coldness as measured on a specific scale.
Desiccants are usually sold by performance (rate, capacity), not by weight. A standard performance definition is the desiccant unit. One unit of desiccant is defined as having an adsorption capacity of 6 grams of water at 77°F (25°C) and 40% RH. For instance, a unit of silica gel weighs 26 grams, a unit of molecular sieve weighs 32 grams and a unit of clay weighs 33 grams.
A space in which the pressure is far below normal atmospheric pressure.
The partial pressure of water vapor in the atmosphere
Water in the form of a gas, especially when below the boiling point and diffused.
The rate that water permeates through the wall or barrier of an enclosure, usually given in g/(m2 x day)or g/(in2 x day).
A thermometer having the bulb covered with a cloth, usually muslin or cambric, saturated with water in which the temperature reading is affected by the amount of humidity in the air.
FAQs
The desiccator cartridge for standard 1″ breather valves can be up to about 18″ long and they will hold about 6 grams of desiccant per inch of length.
However, anything over about 6″ should be supported inside your package as the porous polyethylene tubing is not strong enough to support the cantilevered desiccant weight beyond that. Desiccant cartridges are not able to be attached to the 2″ or 4″ breather valves.
While both are packages intended to hold desiccant, a desiccant basket holds pre-packaged or bagged desiccant. It is intended to mount through a container wall and provide an easy way to replace desiccant from the outside of the container. Our desiccant baskets have long-lasting, easily removable, stainless steel covers that will accept a security wire to provide evidence of tampering and come in a variety of standard lengths. Custom lengths are available upon request.
On the other hand, desiccators are usually packed with loose, or bulk desiccants, and are located inside the container or item. They are intended to keep a specific sealed space dry. Some of our customers’ complex pieces of hardware have had as many as six desiccators, each protecting a different area of the item, as well as a desiccant basket in the container to keep the container interior and item exterior dry.
A breather desiccator is a desiccant cartridge that is mounted to a breather valve. Because the air that passes through the breather valve is forced through the desiccant in the cartridge screwed into the back of the breather valve. With a static desiccator, the air is not forced through the desiccant bed.
Yes. The desiccant cartridge reduces the flow for a given pressure drop by 67 – 75%.
The most common are molecular sieve, either Type 4A or Type 13X, or silica gel. However, any desiccant material that does not “pack” will work. When the desiccant material “packs” it can completely block the flow of air through the desiccator, rendering it non-breathable. It will still function to desiccate, but not for breathing.
AGM has made desiccators in literally hundreds of shapes and sizes ranging from the size of a small grape up to eight foot long tubes and every conceivable size and shape in between. Desiccators have been made from aluminum, stainless steel, bronze and many plastics including polystyrene, ABS, polycarbonate, acrylic, polyester, polyproplene, and combinations of plastics and metals. Manufacturing processes for desiccator components can utilize one or more of the following: milling, turning, stamping, vacuum forming, injection molding and casting.
There are two standard diameters currently available, the 2″ TA476 series and the 4″ TA486 series. Each is available in several standard lengths with custom lengths available upon request.
The TA476/TA478 series require a 2.015″ diameter mounting hole while the TA486 series requires a 4.015″ diameter mounting hole. All necessary mounting hardware is provided.
Find What You're Looking For
It’s a lot, we know. If you’re looking for something specific or aren’t sure what you need, try searching through all products or give us a shout and we’ll help!













